Wow, that is impressive. I on the other hand have been using the Betty Crocker method. I have a nice set of measuring spoons and cups.
Cool thread
Cool thread
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature currently requires accessing the site using the built-in Safari browser.


If you want to get serious, you could always make up your own standard
If I could buy a (reasonably priced) standard, I probably would. But I don't think I would trust a DIY standard any more than I trust the values now.
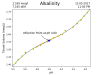
That would probably reduced the fluctuations somewhat. But that is not possible in my sump. I don't have multiple chambers and even if I did, both the tank water and the in-sump filters drain directly into the same filter sock so there is no location in the sump I can pull a sample of water from that has not been contaminated by sump-water.If your readings were sampled one sump chamber upstream from where top off and kalk are added, would that make a difference in stabilizing your readings since the water wouldn't be concentrated or diluted, or not really?
That would probably reduced the fluctuations somewhat. But that is not possible in my sump. I don't have multiple chambers and even if I did, both the tank water and the in-sump filters drain directly into the same filter sock so there is no location in the sump I can pull a sample of water from that has not been contaminated by sump-water.
I have programmed my controller to stop dosing and top-off for 10 minutes prior to each test. That did greatly reduce the fluctuations.
It would only require changing one line of code to have it slowly draw the sample over the course of an hour, then each test would represent the average alk over that hour. That should eliminate evidence of any short term alk fluctuations. But I don't really love that idea. Now that I have convinced myself that the results are accurate, it does not bother me too much to see the fluctuations.
If you are still considering doing a slow sample draw, I would make sure the pH isn't dropping substantially in your sample chamber. It might be overly cautious, but something worth looking at.
That would not be a problem. Changes in pH due to changes in CO2 will not have any impact on the result. Evaporation in the sample chamber would not matter either. The only thing that would be a problem is calcification - and I could pre-dose a bit of HCl to ensure the pH is low enough that there will be no calcification.
However I realized there is a big problem with my slow-draw idea. The pH probe would be out of the water for an hour and that would damage the probe.
There was an interesting debate on that on RC a while ago, but a moot point nonetheless in your case. http://www.reefcentral.com/forums/showthread.php?t=2130613
I could have missed this while reading through this but I had a couple questions regarding your auto-traitor
How is the sample chamber being mixed during the titration? Is there a stiring mechanism? it seems like your titration curves look good so my guess is you are.
Do you ever notice bubbles trapped under by the probe? or considered a smaller probe so you could use a smaller sample size? like these probes, they are an additional cost though.
Have you considered using a Gran Function plot instead of the inflection point? I believe it accounts for sulfates giving a more accurate reading.
I wouldn't expect that you would need to refill super often since the water has a fair bit of ionic strength. I could be way off base however.Eventually I am planning to post the whole project in sufficient detail that anyone could recreate it.
I have a small motor with magnets attached under the reaction chamber and a magnetic stir bar in the chamber.
I have not thought to look for bubbles. Not sure if it would matter though, I seem to get OK results when the probe's junction is just barely touching the sample - and in that case the whole room is a bubble.
I don't feel any need for a smaller sample size. With my current vial (a shotglass) the minimum sample size is 10 ml. With a 10 ml sample it just barely touches the junction so I have been using a 12 ml sample just to be safe(although I get the same results using the 10ml sample). From a tank water consumption side of things, that is plenty small. And if I want to reduce reagent usage I can just use a more potent reagent. Plus, that probe looks really fragile.
I am considering switching to a liquid filled probe instead of a gel filled probe so I can speed up the titration. The main thing holding me back is I can't find any good stats on how often you have to refill the liquid in a liquid probe. Everything I read just says check and refill the probe if needed each time you use it which is not possible since the probe is constantly in use.
I'm embarrassed to say I have no idea what a "Gran Function" is. But you can bet I'll be googling that this evening.
and in that case the whole room is a bubble.
Everything I read just says check and refill the probe if needed each time you use it which is not possible since the probe is constantly in use.
I think this is code that uses a modified gran plot function that doesn't require a calibration. I'm inept when i comes to programming so I don't know if it's usefulI'm embarrassed to say I have no idea what a "Gran Function" is. But you can bet I'll be googling that this evening.
This is because it is dependent on how often the probe is used. The fill hole needs to be opened when it is in use. This allows the fluid to flow through the junction. If the fill hole is closed it can take longer for the reading to stabilize. I'm not sure how long it would take to deplete but I'm guessing if it siting in higher ionic strength solutions, like seawater, it would deplete more quickly. The storage solution for these probes is a KCl solution that is similar to the concentration of the internal probe solution, which prevents fluid transfer between the junction. I think it would be something to check weekly maybe refilling monthly. If you could keep the probe stored in a storage solution it would be even less frequent. Don't store it in RODI.
I think this is code that uses a modified gran plot function that doesn't require a calibration. I'm inept when i comes to programming so I don't know if it's useful
https://water.usgs.gov/owq/FieldManual/Chapter6/section6.6/pdf/6.6.pdf said:Alkalinity, ANC, and concentrations of bicarbonate, carbonate, and hydroxide species are most commonly determined by analyzing acidimetric-titration data with either the inflection point titration method (section 6.6.4.B) or the Gran function plot method (section 6.6.4.C).
► The Inflection Point Titration (IPT) method, also called the incremental titration method, is adequate for most waters and study needs. Difficulty in identifying the inflection points using the IPT method increases as the ratio of organic acids to carbonate species increases and (or) as the alkalinity decreases.
► The Gran function plot (Gran) method is recommended for water in which the alkalinity or ANC is expected to be less than about 0.4 milliequivalents per liter (meq/L) (20 milligrams per liter (mg/L) as CaCO3), or in which conductivity is less than 100 microsiemens per centimeter (µS/cm), or if there are appreciable noncarbonate contributors or measurable concentrations of organic acids.
The Fixed Endpoint method (titration to pH 4.5) rarely is used and is no longer recommended by the USGS for determining alkalinity values because it is less accurate than the IPT and Gran methods. This decrease in accuracy is evident particularly for low concentrations of total carbonate species and for water with significant organic and other noncarbonate contributors to alkalinity or ANC