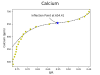
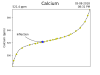
As cypho alluded ( edit- not eluded) to, it's all about the fluctuations. Depending on when the lights turn of and on compared to KHG sampling, I can get a fluctuation of up to about .2 dkH. Technically that is acceptable in the KHG literature as they claim a +- .1 on accuracy.The KHG wants to then dump a bunch of bicarb in and I get some system perturbation.
I ended up using it as mostly a monitor and limited it to being able to add equivalent to a .03 dKH amount per test result (every 4 hours.)
I ended up using it as mostly a monitor and limited it to being able to add equivalent to a .03 dKH amount per test result (every 4 hours.)
Last edited: