Reefer Oz
Keep reefing HARD!
BOD
Lifetime Member
Event Committee
TCMAS Member
TCMAS Supporter
Meeting Host 2021
Meeting Host 2022
Meeting Host 2023
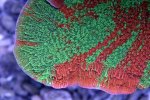
Hungry corals are healthy corals! Looking great dude. Love me some NPS!
How often do you target fed em?
How often do you target fed em?